See more Titanium products. Titanium (atomic symbol: Ti, atomic number: 22) is a Block D, Group 4, Period 4 element with an atomic weight of 47.867. The number of electrons in each of Titanium's shells is [2, 8, 10, 2] and its electron configuration is [Ar] 3d2 4s2.  The titanium atom has a radius of 147 pm and a Van der Waals radius of 187 pm. Titanium was discovered by William Gregor in 1791 and first isolated by Jöns Jakob Berzelius in 1825. In its elemental form, titanium has a silvery grey-white metallic appearance. Titanium's properties are chemically and physically similar to zirconium, both of which have the same number of valence electrons and are in the same group in the periodic table.
The titanium atom has a radius of 147 pm and a Van der Waals radius of 187 pm. Titanium was discovered by William Gregor in 1791 and first isolated by Jöns Jakob Berzelius in 1825. In its elemental form, titanium has a silvery grey-white metallic appearance. Titanium's properties are chemically and physically similar to zirconium, both of which have the same number of valence electrons and are in the same group in the periodic table.  Titanium has five naturally occurring isotopes: 46Ti through 50Ti, with 48Ti being the most abundant (73.8%). Titanium is found in igneous rocks and the sediments derived from them. It is named after the word Titanos, which is Greek for Titans.
Titanium has five naturally occurring isotopes: 46Ti through 50Ti, with 48Ti being the most abundant (73.8%). Titanium is found in igneous rocks and the sediments derived from them. It is named after the word Titanos, which is Greek for Titans.
Materials
Materials by Form
2D Materials Alloy & Alloy Forms Pure Metals & Metal FormsCeramic FibersFoams: Metallic & Ceramic High Purity Materials Isotopes MXenesOxides Rare Earths Semiconductors Solutions
Chemicals & Salts
All Chemicals & Salts Acetates Aluminides Ammonium Sulfates Antimonides Arsenates Benzoate Bromates Bromides Carbonates Chlorides Chromates Fluorides Hydrides Hydroxides Iodates Iodides Lactates Molybdates Nitrates Oxalates Oxides Perchlorates Phosphates Selenates Selenides Selenites Silicates Stearates Sulfates Sulfides Sulfites Tantalates Tellurates Tellurides Tellurites ThiocyanatesVanadates
Ceramics
Nanomaterials
Organometallics
Materials by Application
Additive Manufacturing & 3D Printing Battery & Supercapacitor Materials Catalysts Dental Materials Electronics Materials Fuel Cell Materials Fusion EnergyGlass Manufacturing Green Technology & Alternative Energy Hydrogen Storage Laser Crystals Life Sciences & Biomaterials Metallurgy Nanotechnology & Nanomaterials Optical Materials Photovoltaic & Solar Energy Plating Pigments & Coatings Research & Development Space Technology Sputtering Targets Thin Film Deposition Water Treatment Weather Modification
Life Science Chemicals
Life Science Products AlcoholsAldehydesAmidesAminesAmino Acids & DerivativesAromaticsArylsAzetidinesBenzimidazolesBenzisoxazolesBenzodioxansBenzofuransBenzothiazolesBenzothiophenesBenzoxazolesCarboxylic AcidsEnzymes & InhibitorsEstersEthersFluorinated Building BlocksFuransHalidesImidazolesImidazolidinesIndazolesIndolesIndolinesIsoquinolinesIsoxazolesKetonesMorpholinesNaphthyridinesNitrilesOrganoboronOrganosiliconOxadiazolesOxazolesPharmaceuticals & IntermediatesPhenolsPhytochemicalsPiperazinesPiperidinesPyrazinesPyrazolesPyridazinesPyridinesPyrimidinesPyrrolesPyrrolidinesPyrrolinesQuinazolinesQuinolinesQuinoxalinesSpiroesSulfonyl ChloridesTetrahydroisoquinolinesTetrahydropyransTetrahydroquinolinesTetrazolesThiadiazolesThiazolesThiazolidinesThiolsThiophenesTriazinesTriazoles
About Us
Locations
Austria Belgium Brazil Canada China & Hong Kong Czech Republic Denmark Finland France Germany Greece Hungary India Indonesia Israel Italy Japan Malaysia Mexico Netherlands Norway Philippines Poland Portugal Russia Singapore South Korea Spain Sweden Switzerland Taiwan Thailand Turkey United Kingdom United States
Industries
Aerospace Agriculture Automotive Chemical Manufacturing Defense Dentistry Electronics Energy Storage & Batteries Fine Art Materials Fuel CellsFusion Energy Glass Investment Grade Metals Jewelry & Fashion Lasers Lighting Medical Devices Museums & Galleries Nuclear Energy Oil & Gas Optics Paper & Pulp Pharmaceuticals & Cosmetics Research & Laboratory Robotics Solar Energy Space Sports Equipment Steel & Alloy Producers Textiles & Fabrics Water Treatment Municipalities
Follow Us
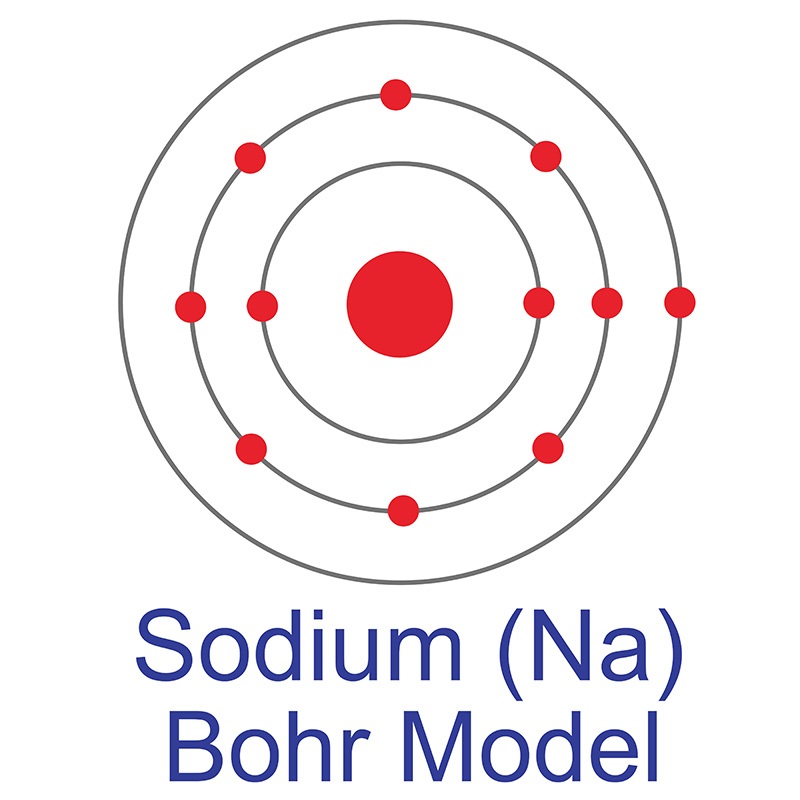 See more Sodium products.
See more Sodium products.